I see a lot of online comments from people who feel their tap water isn’t suitable for plants. People are moving towards distilled, RO water, or rainwater instead. Is this necessary? Is your tap water a problem?
The following are some concerns people have.
- Chlorine
- Fluoride
- pH
- Hardness (high calcium and magnesium) & alkalinity
- Lead
Tap water is rarely a problem for outdoor gardens growing in real soil, but it can be a problem for houseplants and plants growing in soilless mixes, and that is the focus of this post.

Key Takeaways
- Chloride is usually not a problem.
- Hardness and alkalinity can be a problem.
- Lead is usually not a problem.
How to Know What is in Your Tap Water?
The only way to know if your tap water is suitable for plants is to know the amount of various chemicals in your water. If you are on a well, you may need to have your water tested to obtain these values.
If you are on municipal water, you can probably get the data from the city water department. Often, these results are online.
My town produces an annual water report, which is online, and it provides details of the pH and the amounts of various chemicals in our water.
For example, in preparation for this post, I looked up my values and got the following.
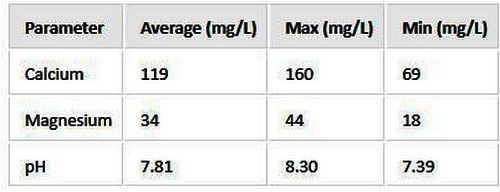
The average values will be close enough for your purposes.
A comment about units is in order. The amounts for chemicals in water are usually reported as mg/L or ppm. If the values are chemicals in solid material, like a bag of fertilizer or soil, they might also be reported as mg/Kg. All of these units are the same mathematically. 10 ppm = 10 mg/L = 10 mg/Kg.
I encourage all gardeners to start using ppm units in their discussions, and I’ve prepared a ppm calculator to help you with this.
Chlorine in Tap Water
I have dealt with this topic in detail in Chlorine, Chloramine and Plants – Everything You Need to Know.
Contrary to popular concerns, neither chlorine nor chloramine from municipal tap water is a concern for plants, provided the World Health Organization Recommendation of less than 5 ppm is followed. A few specialty types of plants may be harmed if the level is above 2 ppm.
The many reports on social media of plants being harmed by chlorination are based on anecdotal information and could be the result of all kinds of issues. The symptoms of chlorine poisoning, such as browning of leaf edges, can be caused by numerous things, including improper watering and overuse of fertilizers. This is a good example of why anecdotal information is of little use in trying to understand the science.
Letting water sit overnight will reduce chlorine, but it does not affect chloramine. However, there is little to no value in doing this since chlorine is not a problem.
Fluoride in Tap Water
When I started writing this article, I was quite sure that the fluoride in tap water would not harm plants, but this may not be true. The answer is certainly much more complex than gardening reports suggest. Here are some key points to understand.
- Plants can be harmed by fluoride without showing any visual symptoms. Gardeners frequently comment, “My water is fluorinated, and my plants are fine”. The problem with this statement is that gardeners rarely do controls, and the absence of symptoms does not mean plants are not harmed. They could be growing more slowly than normal due to fluoride.
- The presence of fluoride toxicity symptoms does not mean they are caused by fluoride. These symptoms can be caused by several other factors, including other salts, low humidity, or even plant shine products.
- Some plants are more sensitive than others.
- The soil or potting media has a significant effect on the amount of fluoride that is absorbed by plants. Clay, organic matter, high levels of calcium, and higher pH all reduce the impact of fluoride on plants.
- Fluoride is accumulated in plants, which means the amount in plants goes up over time. Most testing is short-term, while plants held by gardeners are long-term.
Most of the research I found looked at fluoride levels of 10 ppm or even 100 ppm, while tap water levels are in the order of 1 ppm. I found very little data on the effect of fluoride at 1 ppm and no long-term studies. To be fair, a lot of research on this was done prior to the 1950s and may not be readily available. It is quite possible that low levels of fluoride do not harm plants during the relatively short production/nursery phase, while harming them once grown for several years by gardeners.
The level of fluoride in most tap water, even if it is fluorinated, is about 1 ppm. That level does not seem to cause visible symptoms on most plants. We don’t know if it stunts growth.
pH of Tap Water
The pH of most tap water is suitable for houseplants because they can tolerate a wide range. The ideal range is 5.5–7.0.
More detail:
- Below 5.0: Water is too acidic; it may damage roots and increase the solubility of toxic metals (like aluminum or manganese).
- 5.0–5.5: Acceptable for acid-loving plants (ferns, orchids, African violets, azaleas), but may cause nutrient imbalances for others if used long-term.
- 5.5–6.5: Best range for nutrient availability in soilless mixes.
- 6.5–7.0: Still safe; most houseplants handle this fine.
- Above 7.5: Can cause iron, manganese, and phosphorus deficiencies over time, especially in peat-based mixes.
- Above 8.0: Generally unsuitable for long-term use without acidifying.
Plant roots are fairly insensitive to pH, and they can condition the pH in the rhizosphere to meet their needs. However, alkalinity can be a bigger problem since it buffers the soil and prevents roots from being able to condition the pH. Even a pH of 7.5 is fine for plants, provided the alkalinity is low.
If your pH is high, keep an eye on the alkalinity level.
Hardness & Alkalinity
People commonly confuse the terms harness, alkalinity, calcium and magnesium. Let’s understand the differences.
Calcium and magnesium are two macronutrients that plants need.
Alkalinity is a measure of the total carbonates (CO3), bicarbonates (HCO3), and hydroxyl ions (OH) and is usually expressed as the equivalent of CaCO3, e.g., 100 ppm CaCO3. Although calcium commonly combines with carbonates, alkalinity is not a measure of calcium. I discuss alkalinity in more detail in What is Alkalinity – It May Not Be What You Think?
Hardness is the concentration of divalent cations, mainly calcium (Ca²⁺) and magnesium (Mg²⁺), in water.
I have discussed alkalinity and hardness in detail in Water Hardness and Alkalinity – Why They Both Matter. That article will also help you figure out the values for your tap water.
Both hardness and alkalinity are important. The recommendation is for hardness to be below 200 (100 is even better) and alkalinity below 120.
If your tap water has values above this, you can dilute it with pure water, including RO, distilled water, dehumidifier water, or rainwater.
If your tap water is too pure, it won’t contain enough calcium and magnesium, which are macronutrients for plants. In that case, you need to add them. RO, distilled water, dehumidifier water, or rainwater fall in this category and need to have calcium and magnesium added.
The recommended amounts vary depending on the source of the information; however, the following are the most popular.
- Calcium: 40-120 ppm, with 100 ppm a common suggestion.
- Magnesium: 20-40 ppm, with 25 ppm a common suggestion.
- The ratio of calcium to magnesium is also important and should be in the range of 3-5 ppm calcium per 1 ppm magnesium.
Lead in Tap Water
A number of online sources suggest that lead in tap water can be a problem for houseplants. There are two potential problems: growing food and growing ornamental plants.
Growing Ornamental Plants
The safe lead levels for growing plants are as follows.
- <0.2 ppm: Generally considered safe for most plants. Many guidelines for irrigation water use this as the upper limit.
- 0.2–5 ppm: Some tolerant plants may still grow, but sensitive species can show reduced growth or root injury. The risk of buildup in soil becomes more important.
- >5 mg/L: Considered toxic for most plants — growth and yield are significantly affected.
Most municipalities keep their lead levels in drinking water well below 0.015 ppm. Clearly, such water is not a problem for houseplants.
Growing Food
Lead is a heavy metal that accumulates in our bodies, which means that over time, we have more and more of it in our bodies. That is why it is important to reduce our lead consumption.
The reality is that lead is found everywhere. It exists in all soil, so all vegetables and fruits contain some lead. It is also found in drinking water, although municipalities take steps to keep this level low.
Municipal drinking water contains low levels of lead, such that it is not a problem for growing food. However, some older homes and municipal water delivery systems use lead in their pipes, and this can increase the level. If you suspect lead pipes, have your water tested. If it is safe for drinking, it is safe for growing food.
For information about lead in your soil, see Is Lead in Garden Soil Killing You?




