Mycorrhizal Inoculant products have been around for more than 10 years but the number of products available is rapidly growing. Clearly manufacturers are finding customers willing to pay for these products, but are they worth the money? Do they work? Are companies able to provide solid evidence that their products work?
I have done an investigation and you will be surprised by the results.

Mycorrhizal Inoculant Investigation
If you are not familiar with these products have a look at Mycorrhizae Fungi Inoculant Products.
I have studied the science on these quite a bit, but I wanted to know what data the industry has to support the use of their products. Have they done scientific studies to verify the claims they make?
I Googled for “mycorrhizal inoculants” and picked out 5 brands that seemed to be popular, at least in my Google feed. Each of these products is available from numerous resellers in North America and presumably they are purchased by a lot of people. Each one of them have lots of 5-star reviews – for what that is worth.
To be fair to other manufacturers, I will include their products in the blog post, provided someone adds a comment to this post and includes a link to the manufacture’s scientific supporting research.
The products I selected are:
Pro-Mix – I do use their soil products but not Connect, their mycorrhizal inoculant.
Myke – a product that has been around for many years.
Dnynomyco – a new product with flashy packaging.
Root Rescue – an Ontario, Canada company with strong horticultural family roots.
Plant Revelation Inc – a new company with attractive packaging.
Claims vs Facts
There is a huge problem with this industry and many other similar industries in the way they misuse scientific facts.
There is clear evidence that mycorrhizal fungi in soil provide a lot of benefits to plants. This is not under dispute. About 80% of plants form mycorrhizal connections.
The problem is that companies claim their product has the same benefits as naturally occurring fungi. To the lay person this seems perfectly logical. If fungi in soil help plants, why would adding more not have the same benefits? WHY? For several important reasons.
Firstly, the commercial products might not be viable – dead spores don’t grow. Secondly, they might not be the right microbes for your plants. Thirdly, and most importantly, the soil is already saturated with fungi. Fungi in soil are at capacity. Adding more does not increase the number of living fungi in soil. This last point is explained fully in this video about compost tea – adding it to soil doesn’t work for the same reason.

In order for manufacturers to make efficacy claims about their products, they need to demonstrate that they actually work in the field (ie not in lab pots). For gardeners this means testing in at least agricultural fields.
Online Evidence of Efficacy
Step one in this investigation was to review each website and check for some scientific evidence that their product works. I consider this a bare minimum for any company who wants to sell this kind of product.
None of the 5 manufacturers provided evidence on their website that their product works. For a consumer this should be a big read flag for all 5 mycorrhizal inoculants. If I missed such information, let me know in the comments.
Root Rescue did say, An in-depth, five-year study involving over 21 different species of trees showed that those who formed a symbiotic relationship with the Mycorrhizal Fungi were able to adapt and tolerate the stress and challenges of a dry summer season and still thrive. It is not clear if untreated trees also formed such a relationship, but most importantly they did not provide a link to the research study, so the information is not of much value.
A Second Chance to Prove Efficacy
Websites are made by marketing people who may not understand the importance of verifying the worth of a product, so I gave each company another chance.
I contacted them all and asked for the scientific evidence that supports the claims they make about their product.
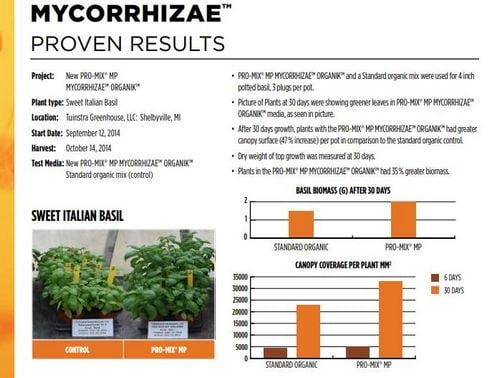
Pro-Mix got back to me right away and provided so-called “scientific evidence” as seen here. This is an internal study that concludes their product works. It has several glaring flaws.
- It was done internally by their researchers – I am sure they are not biased!
- It is not published except as an internal marketing piece.
- No stats were presented for the data – so you can’t reach any conclusions.
- If you look closely, the control soil is completely different than the test soil, so even if we accept the data, it tells us nothing about their inoculant. It only tells us that the two soils are different. Clearly this has not been done by a scientist.
It is a good try, but this is just a marketing piece that does not prove their product works.
Root Rescue also got back to me right away and provided a real scientific research paper that used their product. The website claims that 2,100 trees were tested over 5 years, including 21 species and results show the inoculant “assisted in transplant shock”. The supporting study looked at 6 species between June and Sept of the same year using far fewer trees. The water pressure in the trunks was measured and those growing with inoculant showed higher pressures possibly indicating less drought stress. It is not clear if the drought stress would have occurred in a normal planting with access to water and there is no follow up data to show the long term effects of the inoculant.
Unfortunately the supplied study does not show efficacy of the product.
The other three brands did much worse. They could not even be bothered to respond.
Efficacy of Commercial Products
One study tested 10 commercial mycorrhizal inoculants on a variety of soil-based and soilless potting substrates and found that the “percentage of mycorrhizal colonization obtained with the different mycorrhizal inoculants ranged from 0 to 50%”. This variation was due to both viability of the inoculant as well as the soil itself. “Mycorrhizal colonization did not enhance plant growth”.
Dr. Cobbs, who has done presentations for the Soil Food Wed School, tested commercial fungal products and found, “no evidence of benefit; commercial inoculants never increased native plant biomass, although several inoculants decreased the growth of native species and increased the growth of invasive plant species.”
Similar results have been found in other studies.
Science on Mycorrhizal Inoculants
There are many studies on mycorrhizal inoculants and for the most part they either don’t work or only work in specific cases. They have been shown to work in both drought and high saline conditions but these are usually not a concern for gardeners. None of the manufacturers of these products provide guidelines for using the product in special situations where they might actually provide a benefit. Instead, they recommend them as a general tonic to be used all of the time and science does not support that kind of use.

The Latest Science About These Fungi
The latest science investigates how tilling affects fungi in soil, and looks at efficacy of commercial products in agriculture. Read all about it in Mycorrhizal Fungi – The Latest Scientific News.
Should Gardeners Use Mycorrhizal Inoculants?
Most well tended gardens don’t have plants stressed by drought or saline so the science does not support their use.
Most gardens have lots of mycorrhizal fungi in their soil and these will inoculate the plants on their own.
Gardeners who use a lot of fertilizer should not bother with inoculants since high levels of nutrients inhibit the plants from initiating a connection with fungi.
I have not seen evidence that these products will be helpful for most gardens. The exception might be very dry gardens that don’t have access to enough water.
Better Ways to Build Soil Health.
10 Easy Soil Testing Methods For Measuring Soil Health
Preventing a Nitrogen Deficiency in Soil – How to Manage Nitrogen Levels





It would be interesting to try some experiments with obligate mycorrhizal fungi plants like lavender.
The interactions of mycorrhiza with annual and biennial plants may be different to those of trees and other perennial plants. As a vegetable grower, I am wondering if there is any advantage in adding mycorrhizal fungi to my transplant holes. The disadvantages of competition with native mycorrhizal fungi and distortion of the population already in the soil may lead me to avoid these products. The use of Trichoderma and Bacillus as pest and disease suppressants also worries me. I am wondering if just one dose would give you a population in your soil and no other additions are needed. Whether these preparations work or not may not be the question to be asked. I would rather promote the growth of indigenous by using homemade vegetable mould
Hi Robert,
I read your page with interest, hoping to see comparisons among the various mycorrhizal inoculants on the market. I assume that such data is forthcoming. We would be happy to help you with such a comparison if necessary, though neutrality is key when performing such a trial.
Regarding mycorrhizal saturation – Natural soils are teeming with mycorrhizae, but most agricultural soils are lacking. This is due to modern cultivation methods, which pretty much kill most microbes in your soil. We pride ourselves on bringing life back into your soil – and mycorrhizae are at the heart of any healthy rhizospheric ecosystem.
Regarding scientific data, I wish I could share our brochure herein, but your website allows me to enter only text. Here is a link to a pamphlet that contains some results from external trials: https://workdrive.zoho.com/file/q3jss0b1e6ecc8f45482489ab7d4e6d1234b2. Obviously we have plenty more data, internal and external, which we are happy to share off-line, depending on your needs.
Finally, regarding DYNOMYCO C, this product is being discontinued, in favor of DYNOMYCO (which is now also registered in Canada) and three new products that are in the registration pipeline (Canada is slow with registration). To that extent, I urge you to follow up with trials using DYNOMYCO, rather than DYNOMYCO C. Both are great products, but DYNOMYCO is now the commercial standard. And all of our products are grounded in science! Over thirty years of research have brought us to where we are today. Our website is cannabis-clean, but we have plenty of great testimonials showing that our customers are getting great results with our products – from the individual home grower all the way up to the Master Grower on large farms. Once you grow – you know.
DYNOMYCO Sam
“Natural soils are teeming with mycorrhizae, but most agricultural soils are lacking” – correct – but the reason they are lacking is that the required resources for more growth is not there. Agricultural soil are at capacity. Adding more fungi won’t change that.
“modern cultivation methods, which pretty much kill most microbes in your soil.” – it does harm fungi, but not bacteria.
The link you provided is password protected – I don’t need another account for a tool I don’t use. I have emailed you.
Your summary makes perfect sense if you assume there soil already has a healthy population of VAM fungi and associated plants to feed them via root exudates. If the soil has a history of fungicide and/or fertiliser use then it could be deficient in
VAM species able to colonize new garden plants. Inoculation has worked for me in dead/abused soils that grew cereals intensively. The VAM required may not be everywhere and could take a long time to recolonise after bring destroyed. So iI would suggest they have a role to play in gardens (and definitely in arable fields).
It seems to be a reasonable conclusion, but where is the evidence that “commercial” products work.
And even if they worked in this special case – why are they being sold as a generally good inoculant for every case?
If people want to increase the amount of mycorrhizal fungi in their soil, then the best way to do so is by ensuring the soil is capable of sustaining them in the first place, as throwing these products onto soil bereft of nutrients needed by the fungi will achieve nothing but a lightening of one’s wallet.
Feed the soil: An inch or so of home made compost applied to the surface every year will achieve this.
Alaux, P. L., Zhang, Y., Gilbert, L., and Johnson, D. (2021). Can common mycorrhizal fungal networks be managed to enhance ecosystem functionality? Plants People Planet. 1–12. doi: 10.1002/ppp3.10178
CrossRef Full Text
Andrino, A., Guggenberger, G., Sauheitl, L., Burkart, S., and Boy, J. (2021). Carbon investment into mobilization of mineral and organic phosphorus by arbuscular mycorrhiza. Biol. Fertil. Soils 57, 47–64. doi: 10.1007/s00374-020-01505-5
CrossRef Full Text | Google Scholar
Babikova, Z., Gilbert, L., Bruce, T. J., Birkett, M., Caulfield, J. C., Woodcock, C., et al. (2013). Underground signals carried through common mycelial networks warn neighbouring plants of aphid attack. Ecol. Lett. 16, 835–843. doi: 10.1111/ele.12115
PubMed Abstract | CrossRef Full Text | Google Scholar
Barreto de Novais, C., Pepe, A., Siqueira, J. O., Giovannetti, M., and Sbrana, C. (2017). Compatibility and incompatibility in hyphal anastomosis of arbuscular mycorrhizal fungi. Sci. Agric. 74, 411–416. doi: 10.1590/1678-992x-2016-0243
PubMed Abstract | CrossRef Full Text | Google Scholar
Beiler, K. J., Durall, D. M., Simard, S. W., Maxwell, S. A., and Kretzer, A. M. (2010). Architecture of the wood-wide web: Rhizopogon spp. genets link multiple Douglas-fir cohorts. New Phytol. 185, 543–553. doi: 10.1111/j.1469-8137.2009.03069.x
PubMed Abstract | CrossRef Full Text | Google Scholar
Beiler, K. J., Simard, S. W., and Durall, D. M. (2015). Topology of tree-mycorrhizal fungus interaction networks in xeric and mesic Douglas-fir forests. J. Ecol. 103, 616–628. doi: 10.1111/1365-2745.12387
CrossRef Full Text | Google Scholar
Bever, J. D., Dickie, I. A., Facelli, E., Facelli, J. M., Klironomos, J., Moora, M., et al. (2010). Rooting theories of plant community ecology in microbial interactions. Trends Ecol. Evol. 25, 468–478. doi: 10.1016/j.tree.2010.05.004
PubMed Abstract | CrossRef Full Text | Google Scholar
Bezrutczyk, M., Yang, J., Eom, J. S., Prior, M., Sosso, D., Hartwig, T., et al. (2018). Sugar flux and signaling in plant-microbe interactions. Plant J. 93, 675–685. doi: 10.1111/tpj.13775
PubMed Abstract | CrossRef Full Text | Google Scholar
Bidartondo, M. I., Redecker, D., Hijri, I., Wiemken, A., Bruns, T. D., Domínguez, L., et al. (2002). Epiparasitic plants specialized on arbuscular mycorrhizal fungi. Nature 419, 389–392. doi: 10.1038/nature01054
PubMed Abstract | CrossRef Full Text | Google Scholar
Bingham, M. A., and Simard, S. (2012). Ectomycorrhizal networks of Pseudotsuga menziesii var. glauca trees facilitate establishment of conspecific seedlings under drought. Ecosystems 15, 188–199. doi: 10.1007/s10021-011-9502-2
CrossRef Full Text | Google Scholar
Bingham, M. A., and Simard, S. W. (2011). Do mycorrhizal network benefits to survival and growth of interior Douglas-fir seedlings increase with soil moisture stress? Ecol. Evol. 1, 306–316. doi: 10.1002/ece3.24
PubMed Abstract | CrossRef Full Text | Google Scholar
Booth, M. G., and Hoeksema, J. D. (2010). Mycorrhizal networks counteract competitive effects of canopy trees on seedling survival. Ecology 91, 2294–2302. doi: 10.1890/09-1139.1
PubMed Abstract | CrossRef Full Text | Google Scholar
Bravo, A., Brands, M., Wewer, V., Dörmann, P., and Harrison, M. J. (2017). Arbuscular mycorrhiza-specific enzymes FatM and RAM 2 fine-tune lipid biosynthesis to promote development of arbuscular mycorrhiza. New Phytol. 214, 1631–1645. doi: 10.1111/nph.14533
PubMed Abstract | CrossRef Full Text | Google Scholar
Breuillin-Sessoms, F., Floss, D. S., Gomez, S. K., Pumplin, N., Ding, Y., Levesque-Tremblay, V., et al. (2015). Suppression of arbuscule degeneration in Medicago truncatula phosphate transporter4 mutants is dependent on the ammonium transporter 2 family protein AMT2; 3. Plant Cell 27, 1352–1366. doi: 10.1105/tpc.114.131144
PubMed Abstract | CrossRef Full Text | Google Scholar
Brundrett, M. C. (2009). Mycorrhizal associations and other means of nutrition of vascular plants: understanding the global diversity of host plants by resolving conflicting information and developing reliable means of diagnosis. Plant Soil 320, 37–77. doi: 10.1007/s11104-008-9877-9
CrossRef Full Text | Google Scholar
Brundrett, M. C., and Tedersoo, L. (2018). Evolutionary history of mycorrhizal symbioses and global host plant diversity. New Phytol. 220, 1108–1115. doi: 10.1111/nph.14976
PubMed Abstract | CrossRef Full Text | Google Scholar
Bücking, H., and Kafle, A. (2015). Role of arbuscular mycorrhizal fungi in the nitrogen uptake of plants: current knowledge and research gaps. Agronomy 5, 587–612. doi: 10.3390/agronomy5040587
CrossRef Full Text | Google Scholar
Bücking, H., Mensah, J. A., and Fellbaum, C. R. (2016). Common mycorrhizal networks and their effect on the bargaining power of the fungal partner in the arbuscular mycorrhizal symbiosis. Commun. Integr. Biol. 9:e1107684. doi: 10.1080/19420889.2015.1107684
PubMed Abstract | CrossRef Full Text | Google Scholar
Burke, D. J., Klenkar, M. K., and Medeiros, J. S. (2018). Mycorrhizal network connections, water reduction, and neighboring plant species differentially impact seedling performance of two forest wildflowers. Int. J. Plant Sci. 179, 314–324. doi: 10.1086/696686
CrossRef Full Text | Google Scholar
Chapagain, T., and Riseman, A. (2014). Barley-pea intercropping: effects on land productivity, carbon and nitrogen transformations. Field Crops Res. 166, 18–25. doi: 10.1016/j.fcr.2014.06.014
CrossRef Full Text | Google Scholar
Courty, P. E., Buée, M., Diedhiou, A. G., Frey-Klett, P., Le Tacon, F., Rineau, F., et al. (2010). The role of ectomycorrhizal communities in forest ecosystem processes: new perspectives and emerging concepts. Soil Biol. Biochem. 42, 679–698. doi: 10.1016/j.soilbio.2009.12.006
CrossRef Full Text | Google Scholar
Deslippe, J. R., and Simard, S. W. (2011). Below-ground carbon transfer among Betula nana may increase with warming in Arctic tundra. New Phytol. 192, 689–698. doi: 10.1111/j.1469-8137.2011.03835.x
PubMed Abstract | CrossRef Full Text | Google Scholar
Diédhiou, A. G., Selosse, M. A., Galiana, A., Diabaté, M., Dreyfus, B., Bâ, A. M., et al. (2010). Multi-host ectomycorrhizal fungi are predominant in a Guinean tropical rainforest and shared between canopy trees and seedlings. Environ. Microbiol. 12, 2219–2232. doi: 10.1111/j.1462-2920.2010.02183.x
PubMed Abstract | CrossRef Full Text | Google Scholar
Ekblad, A., Wallander, H., Godbold, D. L., Cruz, C., Johnson, D., Baldrian, P., et al. (2013). The production and turnover of extramatrical mycelium of ectomycorrhizal fungi in forest soils: role in carbon cycling. Plant Soil 366, 1–27. doi: 10.1007/s11104-013-1630-3
CrossRef Full Text | Google Scholar
Fang, L., He, X., Zhang, X., Yang, Y., Liu, R., Shi, S., et al. (2021). A small amount of nitrogen transfer from White Clover to Citrus seedling via common arbuscular mycorrhizal networks. Agronomy 11:32. doi: 10.3390/agronomy11010032
CrossRef Full Text | Google Scholar
Fellbaum, C. R., Gachomo, E. W., Beesetty, Y., Choudhari, S., Strahan, G. D., Pfeffer, P. E., et al. (2012). Carbon availability triggers fungal nitrogen uptake and transport in arbuscular mycorrhizal symbiosis. Proc. Nat. Acad. Sci. U.S.A. 109, 2666–2671. doi: 10.1073/pnas.1118650109
PubMed Abstract | CrossRef Full Text | Google Scholar
Fellbaum, C. R., Mensah, J. A., Cloos, A. J., Strahan, G. E., Pfeffer, P. E., Kiers, E. T., et al. (2014). Fungal nutrient allocation in common mycorrhizal networks is regulated by the carbon source strength of individual host plants. New Phytol. 203, 646–656. doi: 10.1111/nph.12827
PubMed Abstract | CrossRef Full Text | Google Scholar
Fernandez, M., Malagoli, P., Vernay, A., Ameglio, T., and Balandier, P. (2020). Below-ground nitrogen transfer from oak seedlings facilitates Molinia growth: 15 N pulse-chase labelling. Plant Soil 423, 59–85. doi: 10.1007/s11104-020-04473-9
CrossRef Full Text | Google Scholar
Fitter, A. H., Graves, J. D., Watkins, N. K., Robinson, D., and Scrimgeour, C. (1998). Carbon transfer between plants and its control in networks of arbuscular mycorrhizas. Funct. Ecol. 12, 406–412. doi: 10.1046/j.1365-2435.1998.00206.x
PubMed Abstract | CrossRef Full Text | Google Scholar
Gilbert, L., and Johnson, D. (2017). Plant-plant communication through common mycorrhizal networks. Adv. Bot. Res. 82, 83–97. doi: 10.1016/bs.abr.2016.09.001
PubMed Abstract | CrossRef Full Text | Google Scholar
Giovannetti, M., Fortuna, P., Citernesi, A. S., Morini, S., and Nuti, M. P. (2001). The occurrence of anastomosis formation and nuclear exchange in intact arbuscular mycorrhizal networks. New Phytol. 151, 717–724. doi: 10.1046/j.0028-646x.2001.00216.x
PubMed Abstract | CrossRef Full Text | Google Scholar
Girlanda, M., Selosse, M. A., Cafasso, D., Brilli, F., Delfine, S., Fabbian, R., et al. (2006). Inefficient photosynthesis in the Mediterranean orchid Limodorum abortivum is mirrored by specific association to ectomycorrhizal Russulaceae. Mol. Ecol. 15, 491–504. doi: 10.1111/j.1365-294X.2005.02770.x
PubMed Abstract | CrossRef Full Text | Google Scholar
Gorzelak, M. A., Asay, A. K., Pickles, B. J., and Simard, S. W. (2015). Inter-plant communication through mycorrhizal networks mediates complex adaptive behaviour in plant communities. AoB Plants 7:plv050. doi: 10.1093/aobpla/plv050
PubMed Abstract | CrossRef Full Text | Google Scholar
Grove, S., Saarman, N. P., Gilbert, G. S., Faircloth, B., Haubensak, K. A., and Parker, I. M. (2019). Ectomycorrhizas and tree seedling establishment are strongly influenced by forest edge proximity but not soil inoculum. Ecol. Appl. 29:e01867. doi: 10.1002/eap.1867
PubMed Abstract | CrossRef Full Text | Google Scholar
Gyuricza, V., Thiry, Y., Wannijn, J., Declerck, S., and Dupré de Boulois, H. (2010). Radiocesium transfer between Medicago truncatula plants via a common mycorrhizal network. Environ. Microbiol. 12, 2180–2189. doi: 10.1111/j.1462-2920.2009.02118.x
PubMed Abstract | CrossRef Full Text | Google Scholar
He, X., Critchley, C., Ng, H., and Bledsoe, C. (2005). Nodulated N2-fixing Casuarina cunninghamiana is the sink for net N transfer from non-N2-fixing Eucalyptus maculata via an ectomycorrhizal fungus Pisolithus sp. using 15NH4+ or 15NO3– supplied as ammonium nitrate. New Phytol. 167, 897–912. doi: 10.1111/j.1469-8137.2005.01437.x
PubMed Abstract | CrossRef Full Text | Google Scholar
He, X., Xu, M., Qiu, G. Y., and Zhou, J. (2009). Use of 15N stable isotope to quantify nitrogen transfer between mycorrhizal plants. J. Plant Ecol. 2, 107–118. doi: 10.1093/jpe/rtp015
CrossRef Full Text | Google Scholar
He, Y., Cornelissen, J. H., Wang, P., Dong, M., and Ou, J. (2019). Nitrogen transfer from one plant to another depends on plant biomass production between conspecific and heterospecific species via a common arbuscular mycorrhizal network. Environ. Sci. Pollut. Res. 26, 8828–8837. doi: 10.1007/s11356-019-04385-x
PubMed Abstract | CrossRef Full Text | Google Scholar
Heaton, L., Obara, B., Grau, V., Jones, N., Nakagaki, T., Boddy, L., et al. (2012). Analysis of fungal networks. Fungal Biol. Rev. 26, 12–29. doi: 10.1016/j.fbr.2012.02.001
Nice list of references, but ……
1) You did not provide links for any of them.
2) You did not say what these links are for.
Show me one link that tests a commercial product and shows it works in a non-stressed condition.
Years ago, I wrote to the Pro Mix folks and several horticulture magazine authors who wrote about the benefits of micorrhizal additives. I wanted to know the survival rates of inoculants when bales are stored 5+ months, sometimes at minus 40 temps. This of course is after they have been stored for ??? Weeks/months at the processing facility then transported by truck/ barge to my local supplier. No one answered. Here there is no choice. Pro mix only comes with the inoculant. You can’t do comparison research (and I’m a university scientist). All I know is the price per bale skyrocketed.
You made me grin.
I was at the organic conference Sunday and asked them the same thing – I do use the product. Do they have a study to compare Pro-mix to sterilized pro-mix? The people in the booth didn’t know but they are looking into it for me. Don’t hold your breath.
A comparison between out of the bag and the same material sterilized should give a very clear indication of the amount of surviving fungi.
I doubt the added mycorrhizal fungi have any effect in those bails.
You raise very good points. However, I live in Southwest NM which presents some tough growing conditions, particularly for veggies. Geoff Lawton (of permaculture fame) posted a video years ago of several old CCC(Civilian Conservation Corp) berms built south of Tucson in the 30s. The contrast was striking: outside the berm area we see typical Sonoran Desert vegetation, but inside the catchment area lives a whole different and lush ecosystem. The point is that the mciroorganisms in the catchment area had a longer period to do their thing due to the moisture. The few tests done were probably given consistent moisture, so I wonder how a real world situation such as mine would fare.
Robert, what about using mycorrhizal innoculants in potting soil for containerized plants?
Manufactures of the product don’t have evidence that works.
The issue in pots is that we tend to fertilize the pots. When plants get enough nutrients they won’t make associations with fungi which then die.
Your study would indeed have surprised me – if it had shown these products to be worthwhile! It is such a shame that our growing understanding of the beneficial relationships that plants and animals have with a whole range of fungi, bacteria and archaea has been hijacked by marketers to sell yet more useless products. A similar example is ‘live’ yoghurts (which nowadays normally aren’t even genuine live yoghurts with a range of micro-organisms, but sterilised yoghurts with one or two bacterial strains added back in afterwards). These might be useful after you have killed off your gut flora with a course of antibiotics, but with a normal, healthy gut it is like throwing tree seedlings into a forest from a helicopter.
It’s really a matter of reasoning from first principles: mycorrhizal connections are between specific species of plants and fungi, so any product that’s pitched as having more general benefits is, a priori, a scam.